Background: Donor T cells specific for minor histocompatibility (H) antigens can deliver potent, selective anti-leukemic effects after allogeneic HCT when the antigen is negligibly or not expressed by non-hematopoietic tissues, not present in the donor, and expressed by the recipient. We reported a new minor H antigen-directed T-cell therapy that can be deployed after HCT to manage persistent or recurrent measurable residual hematologic malignancies or overt relapse (Blood 2018;131(1):108). We developed a transgene with 4 components: 1) a high-affinity T-cell receptor (TCR) specific for the hematopoietic-restricted minor H antigen, HA-1 that is presented on HLA-A*02:01; 2) a CD8 co-receptor to enhance function of the class I-restricted TCR in CD4+ T cells so they promote cytotoxic CD8+ T cell function and survival; 3) an inducible caspase-9 safety switch, which can be triggered by the drug rimiducid in case of in vivo toxicity; and 4) a CD34-CD20 epitope to facilitate selection of the engineered product during manufacturing and track HA-1 TCR T cells in the recipient. The 21-day manufacturing process entails CD45RA+ naïve T cell depletion (minimizes the risk of GvHD), and subsequent CD4+ and CD8+ separation (provides a consistent 1:1 CD4:CD8 ratio). The separate cultures are transduced with the lentivirus construct iCasp9-HA1-TCR2-RQR-CD8, expanded, and selected using the CD34 marker to ensure removal of untransduced T cells.
Study Design and Methods: The single-center phase I trial (NCT03326921) evaluates the feasibility and safety of infusion of HA-1 TCR T-cell immunotherapy. Primary end points are 1) Feasibility of manufacturing and administering HA-1 TCR CD8+ and CD4+ memory T cells and 2) Dose-limiting toxicity of HA-1 TCR T cells. Major inclusion criteria are: HLA-A*02:01-positive, HA-1-positive patients who underwent HCT for acute leukemia, myelodysplastic syndrome, BPDCN, CML, CMML or JMML from a HLA-A*-02:01+/HA-1-negative donor or HLA-A*02:01-negative haploidentical or mismatched donor (excluding umbilical cord). HA-1 genotype screening is performed on patient and donor blood, hair follicle or cheek swab samples shipped to Fred Hutchinson Cancer Research Center. To be eligible for treatment, patients must develop measurable residual disease or overt relapse after HCT but may receive other standard or investigational therapies prior to treatment with HA-1 TCR T-cell immunotherapy if clinically indicated. Some systemic immunosuppression may be continued, but prior grade IV acute GVHD and prior severe chronic GVHD are key exclusions. Two groups, <16 and ≥16 years, will be treated at dose levels ranging from 3 x 106 to30 x 106 cells/kg, in cohorts of 3-6 subjects, up to approximately 24 subjects in total. Fludarabine lymphodepletion will be used in most subjects, followed by a single T-cell infusion, with an option for a subsequent infusion(s) if the subject demonstrates an initial response without severe toxicity. Bone marrow aspirations are performed prior to T-cell infusion and at several time points following infusion.
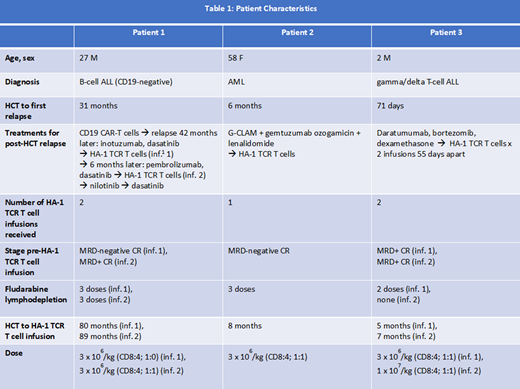
Recruitment and Patient Characteristics: To date, 3 subjects have been treated on the phase I clinical trial and received a total of 5 infusions (Table 1). HA-1 TCR T cell persistence in blood and bone marrow has been documented from >3 months to >13 months. Clear in vivo anti-leukemic activity was observed at the first dose level, including in a subject with aggressive, highly refractory T-ALL and early post-HCT relapse.
Outlook: Minor H antigen-specific T-cell immunotherapy may offer effective management of post-HCT relapse while avoiding GvHD and other off-target effects. Due to population genetics of HA-1 and HLA-A*02:01, HA-1 TCR T-cell immunotherapy is applicable to 10-15% of HCT recipients with various hematological malignancies. The ongoing phase I trial is actively recruiting patients. Development of T-cell immunotherapy targeting other minor H antigen/HLA combinations is also underway to increase the broad applicability of minor H antigen-targeted T-cell immunotherapy.
Krakow:HighPass Bio: Research Funding. Cunningham:HighPass Bio: Research Funding. Vartanian:HighPass Bio: Research Funding. Bleakley:HighPass Bio: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal